29
03/2022
Should children and adolescents be vaccinated against COVID-19 or not? From what age is it safe to immunize minors?
Dr. Oscar Tamez, pediatric infectious disease specialist and member of the TecSalud COVID team, answered for CONECTA the most common questions about vaccination in minors.
“Although adolescents and children do not usually get seriously ill, they are capable of contagious. It is important to vaccinate this population, which is still vulnerable and can also perpetuate the cycle of infection and pandemic. ", He said.
At CONECTA we share answers about the application of anti-COVID vaccines in minors:
Should minors be vaccinated against COVID-19 or not?
The TecSalud specialist pointed out that vaccination in minors should be done to minimize the risk of hospitalization or death.
"In the United States, with this third wave, there was an increase of more than 10 times the number of hospitalizations in pediatric ages, a situation that we had not seen in previous waves.
"As the pediatric population becomes vaccinated, the risk of hospitalization or death in children and adolescents is going to decrease dramatically," he said.
According to a study by the Latin American Center for Journalistic Research, cited by the Animal Politico page, hospitalizations for COVID in children under 18 in Mexico increased 110% between January and August.
The expert referred to these increases due to the fact that this population sector has not yet received the vaccine for the most part, due to the relaxation of measures in summer, as well as due to the emergence of the delta variant.
According to the federal government, minors without chronic diseases are not a population at risk, since deaths from COVID-19 have a low percentage compared to cases. For example, from January to July, of 73,773 cases, 185 have died, that is, 0.25%.
Dr. Tamez adds that another reason to vaccinate children is to avoid so-called prolonged COVID or Long COVID, which is when symptoms of COVID infection persist beyond 4 weeks after primary infection.
"When the authorities give the go-ahead to use these biologicals, without a doubt, all of us must apply them, also to minors, from the ages that are authorized," said the TecSalud doctor.
Pfizer vaccine, the only one authorized for children between 12 and 17 years old
In Mexico, like the United States, so far only the Pfizer-BioNTech two-dose messenger RNA vaccine is authorized for use in children over 12 years of age.
This September 20, Pfizer reported that the vaccine had good results in children aged 5 to 11 years in its studies, so it is preparing to process permits to start vaccination in children of these ages.
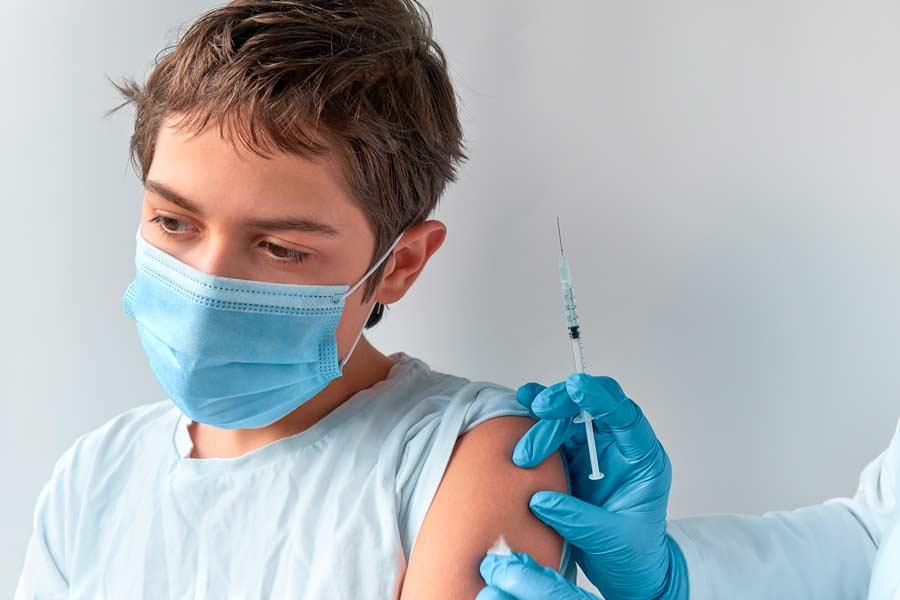
“These are very solid and very encouraging preliminary reports regarding the immune response of people between 5 and 11 years of age. What is most interesting is that it is achieved with a third of the dose of what is usually administered to an adult,” said Dr. Tamez.
Dr. Tamez says that in the United States Moderna's messenger RNA vaccine is in its clinical trials in the recruitment and administration phase for children under 12 years of age.
“This has to be done in stages. They have to go little by little seeing what are the ages that are tolerating the vaccines. For example, results from Pfizer, in the under-5 phase, are expected to be available later this year,” he added.
Mothers can "vaccinate" their babies in their womb
A study in the American Journal of Obstetrics & Gynecology-Maternal Fetal Medicine, published on September 21, revealed that babies of pregnant women who received messenger RNA vaccines are born with high levels of antibodies.
However, the specialist stressed that the Centers for Disease Control and Prevention (CDC) and health organizations still do not recommend using vaccines in babies.
“Once they are already authorized and we have the information available regarding safety and effectiveness, go ahead. Probably a model very similar to what happens with influenza can be taken; vaccinate from 6 months to one year of age.
A4: Just learned how to create a 'Wordle' last week. A teacher could ask the class a question and everyone could go… https://t.co/B3VZOK3l6o
— F.ChristinePowell Fri Oct 16 02:33:58 +0000 2020
Are minors going to get sick from receiving the vaccine?
No person, including minors, will get COVID-19 from receiving any of the anti-COVID vaccines, including Pfizer.
Minors may have side effects such as soreness, redness, and swelling in the arm where the shot was given. In the rest of the body they may have tiredness, headache or muscle pain, chills, fever or nausea.
These effects should wear off within a few days. Some minors have no side effects.
After the injection, the injection site should be left for 15 to 30 minutes to be observed in case a severe allergic reaction occurs and immediate treatment is needed.
Can they have major side effects?
The health specialist pointed out that there is a possibility of having major adverse effects, however, these are not of sufficient risk to dismiss the safety of the vaccine.
“The situation with minors in Pfizer and Moderna clinical trials, in adolescents and pre-adolescents, an adverse effect has been observed in a very small percentage that has not been as observed in adult populations.
“It is called myocarditis and pericarditis. This speaks of inflammation of the layer that covers the heart and also a type of inflammation of the heart muscles, but that is self-limited, that is, it is removed alone or with an anti-inflammatory.
In children under 12 years of age, there are still no results regarding adverse effects, however, reports from Pfizer indicate that the vaccination is safe, the doctor clarified.
Is the same dose applied as for an adult?
In minors, the same vaccine is used as in an adult, but in a lower dose, something that, according to the doctor, has been implemented for other diseases such as influenza, since pediatric patients require less stimulation for a good immune response.
“At least in the studies with Pfizer they were based on two doses separated by 28 days. The dose is 10 micrograms, contrary to what is inoculated in an adult, which is 30 micrograms.
In addition, the vaccine dosage does not vary based on patient weight, as there are no weight requirements for pediatric patients to be vaccinated, according to the CDC.
If a child got sick with COVID, how long does it take to get them vaccinated?
Dr. Tamez noted that minors who have had COVID can be vaccinated after 14 days from the start of symptoms.
"This is with the intention of being outside the contagious period and not generating a risk of contagion to other people," he explained.
Are these tests that are done on vaccines for minors reliable?
“In this type of trial, what they do is inoculate the study subjects with the vaccine or biological, in this case, between 5 and 11 years old,” explained Dr. Tamez.
"What they did was measure the amount of neutralizing antibodies at one and two months and in this case these positive results of protection, which is called immunogenicity, showed practically identical results to those obtained in adults," he added.
What is the difference between anti-COVID vaccines and those for other diseases?
The TecSalud pediatric infectologist pointed out that the main differentiator of anti-COVID vaccines lies in the type of technology used by Pfizer's messenger RNA vaccines.
“On the messenger RNA platform, it is one of the first widely distributed vaccines; its technology can eventually be adopted for other types of diseases. Another difference is their versatility because they are faster to upgrade if the virus mutates,” he added.
Vaccination in Mexico for children with risk factors
This September 24, Hugo López-Gatell, Undersecretary of Prevention and Health Promotion, said that on October 1 the vaccination registry begins with the Pfizer-BioNTech biological for minors who have higher risk factors to be vaccinated.
It is estimated that there are between 750,000 and up to 1,500,000 minors with chronic diseases in Mexico. Your registration will be done through the My Vacuna website.
“These are steps in the right direction and it means that the energy and the strategies have their eyes on the pediatric population,” said Dr. Tamez.
Although it is yet to be officially defined, in Mexico minors will be immunized first with:
On the other hand, some minors in Mexico, without necessarily having any risk factor, have already been vaccinated.
This has been possible thanks to judicial protections or vaccination strategies such as that of the Governor-elect of Nuevo León, Samuel García, of taking the children of workers between the ages of 12 and 17 in buses to be vaccinated at the border with Texas.
With information from Susana Irais.
READ ALSO:

- 1073
- the texas women's health program counts as insurance
Related Articles
How many sit-ups should I do a day to get rid of my belly?
30/01/2022Flattening the abdomen or reducing the fat in this area is a difficult task. But don't despair, we will solve the question: how many sit-ups should I do a day to eliminate the belly. Abdominal fat...
Jalisco The Ministry of Health invites men and women to learn about family planning services
04/02/2022The Jalisco Ministry of Health invites men and women of reproductive age to learn about and use the various services offered by the Family Planning and Contraception Program, which...
Telva International day against gender violence: screens multiply mistreatment against women
01/02/2022SaludUpdated Change of scenery, same victims. On the International Day for the Elimination of Violence against Women, we highlight how aggressions have been copied to the digital sphere, increasing...
Coronavirus Spain today | | Health The Trust Project
18/03/2022The Ministry of Health has reported 3,261 new coronavirus infections and 155 deaths, while the cumulative incidence drops more than six points in 24 hours to 109.3 cases. There is...